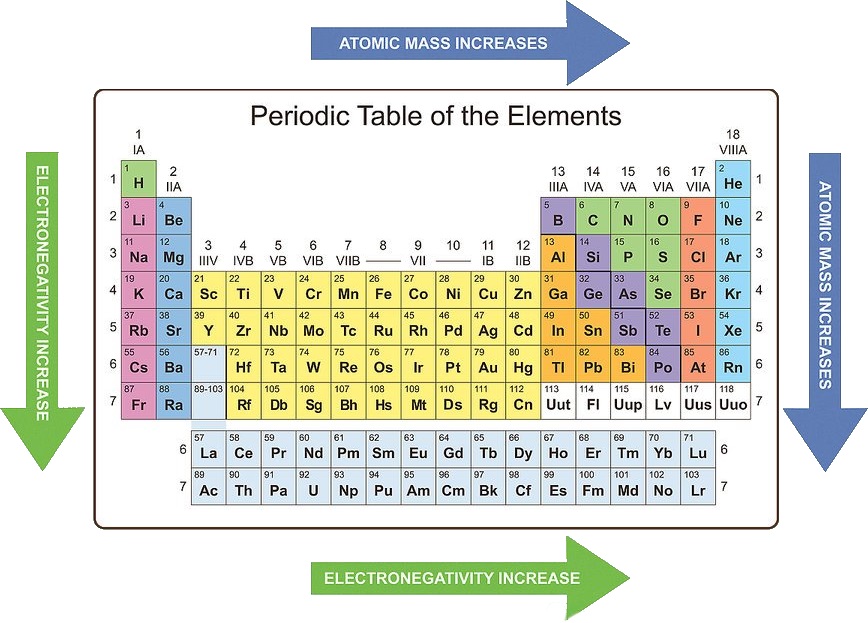
Meyer formed his periodic law based on the atomic volume or molar volume, which is the atomic mass divided by the density in solid form. There is no other tool in science that allows us to judge relative properties of a class of objects like this, which makes the periodic table a very useful tool. They both arranged the elements by their mass and proposed that certain properties periodically reoccur. The variations of properties according to positions on the periodic table are called periodic trends. Mendeleev created the first periodic table and was shortly followed by Meyer. Term Definition Trend in the Periodic Table Why Atomic Radius. The Periodic Law The periodic law was developed independently by Dmitri Mendeleev and Lothar Meyer in 1869. Topic 5 The Chemical Equation Topic 4 Intro to Covalent Bonding.Table Basics The periodic table of elements is one of the ways that scientists keep track of all the known elements. In using this model to describe the electronic structures of the elements in order of increasing atomic number, we saw that periodic similarities in electron.When a neutral atom gains or loses an electron, creating an anion or cation, the atom's radius increases or decreases, respectively. Neutral atoms tend to increase in size down a group and decrease across a period. One such trend is closely linked to atomic radii - ionic radii. Common periodic trends include those in ionization energy, atomic radius, and electron affinity. Periodic Trends in Ionic Radii An understanding of periodic trends is necessary when analyzing and predicting molecular properties and interactions.Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool. Properties that show periodic trends include. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Periodic trends are repeating patterns found in the periodic table as you move across a period or down a group. Since the orbitals around an atom are defined in terms of a. 
Periodic Trends Page notifications Off Share Table of contents Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. The atomic radius is the distance from the nucleus of an atom to the outermost electrons.Understanding these trends is done by analyzing the elements electron configuration all elements prefer an octet formation and will gain or lose electrons to form that stable configuration. All of these elements display several other trends and we can use the periodic law and table formation to predict their chemical, physical, and atomic properties. Definitions and important concepts (ESABN) Atomic radius Ionisation energy Electron affinity Electronegativity A group is a vertical column in the periodic. Periodic Properties of the Elements The elements in the periodic table are arranged in order of increasing atomic number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
